ARTICLE AND PHOTOS BY BRYAN KUDISCH | SCIENCE ISSUE 2015
President of the Chandler Society and chemist Bryan turns to science to find out why we can’t use Splenda™, that much-hyped, zero-calorie sugar substitute, in our baking.
• • • • • • • • • • • • • • • • • • • • • • • • • • • • • • • • •
Even amateur bakers (like me) know that you can’t bake with Splenda™, a sugar substitute which has become more and more common among calorie-counters all over the world. One yellow packet of the stuff boasts zero calories with arguably comparable sweetness to normal sugar, leaving consumers a little bit less guilty as they add some to their iced tea at a restaurant. So why can’t we use Splenda for baking? Well, let’s take a look:
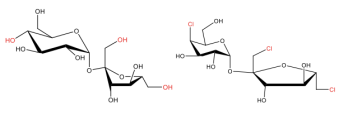
Left: We have the 3D chemical structure of sucrose, or table sugar. Right: We have the 3D chemical structure of sucralose, the ingredient which makes Splenda™ sweet.
So if you look at the two molecules above, all that has changed from going to table sugar to sucralose is a few chlorine atoms replacing a few hydroxyl (-OH) groups, as well as some conformational changes, like the chlorine atom on the left sticking straight up instead of to the side in sucrose. In fact, it is the stereochemistry of sucralose which prevents it from binding to enzymes in your cells; it passes straight through your body without being digested, hence zero calories!
Investigating further into the baking conundrum, a short description of what happens to sugar when it is baked is definitely appropriate:
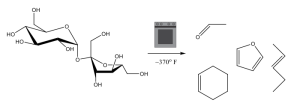
When sugar is placed into a sufficiently high heat oven (the observant reader may recognize a familiar baking temperature), it caramelizes, meaning that it releases some oxygen and forms a class of organic molecules called alkenes. These molecules give caramel its characteristic nutty smell. The way that the caramel turns brown is a bit out of the scope of this article, but interested readers can learn more about the process here.
If you are like me, then this seems like a smoking gun. Even though sucralose might look the same, those chlorine atoms probably change the ability for the molecule to break down when heated, preventing it from being caramelized. Well, you’re not wrong, but actually recent research has found that sucralose caramelizes at a lower temperature than normal table sugar. What gives, Splenda™? Why can’t I bake with you?
It turns out the real reason comes from the fact that sucralose is very sweet. Like, very very sweet—there are estimates that sucralose is almost 1000 times sweeter than normal table sugar. So why aren’t those little yellow packets overpoweringly saccharine? Let’s turn to the ingredient list:
Splenda™ is “diluted,” as chemists say, with other low calorie carbohydrate fillers; namely dextrose and maltodextrin. Maltodextrin is our real smoking gun—its polymer-esque chemical structure gives it a decomposition temperature which is much higher than normal baking temperatures. Hence, even though the sucralose in the Splenda™ might caramelize, there is so little of it in the actual packet that it almost looks like nothing caramelizes at all.
With the mystery solved, it is worth noting that Splenda™ has actually released new variants of its product specifically for baking. They most likely adjusted the ratio of actual sweetener to filler in their recipe in order to allow that nice, caramel flavor that we all love in our baked goods!